Three university juniors joined forces to establish Mezoo in 2007.
For a decade, they secured research funding by developing products for other companies.
They resisted the temptation of incentives tied to relocating to Seoul and chose Wonju instead.
After proving their technological capabilities, they attracted a series of large-scale investments.
Their flagship product is “HiCardi,” a mobile remote patient monitoring solution.
“Next month’s KOSDAQ listing will be a stepping stone toward becoming a global company.”
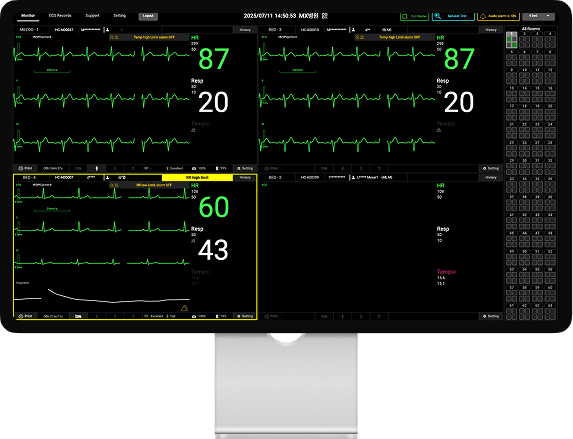
▲ Mezoo enables simultaneous monitoring of multiple patients, allowing up to 256 beds to be viewed on a single screen.
“We completed our undergraduate studies as well as our master’s and doctoral degrees in Wonju, and even started our company here. Investors told us that success required moving to Seoul, but we wanted to prove that a global company can emerge from Wonju as well.”
These are the words of Park Jeong-hwan, CEO of Mezoo (MEZOO), a digital healthcare company based in Wonju that is drawing growing attention. Founded in 2008 in a corner of a research lab at Yonsei University’s Mirae Campus, Mezoo has grown over the past 20 years into a leading company in the healthcare market and a representative enterprise of Wonju. With its KOSDAQ listing scheduled for March this year, the company is preparing for overseas expansion.

▲ Exterior view of Mezoo’s Wonju office
A Dream of a “Zoo” Filled with Medical Devices
Park Jeong-hwan, a member of the Class of ’93 in the Department of Biomedical Electronics Engineering at Yonsei University’s Mirae Campus, founded Mezoo in 2007 together with three junior colleagues. The name Mezoo is an abbreviation of “Medical Equipment Zoo,” reflecting the company’s bold ambition to create all kinds of medical devices with its own hands, just as a zoo houses a wide variety of animals.
Reality, however, was harsh. Although they had strong technological capabilities, they lacked funding. Until 2017, the team endured for nearly a decade relying solely on their technology. To secure research funds, they taught university courses and took on contract development work for other companies. They also earned recognition for their technical expertise by participating for five years in a project to improve the performance of blood glucose meters for I-SENS, a local Wonju-based company.
CEO Park explained, “I decided to start a company because I wanted to bridge the gap between theory and practice, but in the early days, we spent more time developing products for others just to survive.” He added, “Through that process, we were able to accumulate core technologies across a wide range of medical devices, including electrocardiographs, oxygen saturation monitors, and non-contact biometric measurement technologies.”
Source: https://www.kado.net/news/articleView.html?idxno=2033762
